 +86-592-5131 569
+86-592-5131 569info@esep.co
Green technology for sustainable development
WATER SPLITTING
Bipolar Membrane is composed of one cation-exchange layer and one anion-exchange layer joined together. Bipolar Electrodialysis is an ion exchange membrane process that uses a bipolar membrane to split water into H+ and OH-, generating acid and caustic streams. It also is an electrodialysis process since ion exchange membranes are used to separate ionic species in solution with the driving force of an electrical field, but it is different by the unique “water splitting” capability of the bipolar membrane. In addition, the process offers unique opportunities to directly acidify or basify process streams without adding acids or bases, avoiding by-product or waste streams and costly downstream purification steps.
The Bipolar Membrane
A good bipolar membrane has a strong, permanent bond between the two layers and a thin interface to reduce the voltage drop. It also allows the water to easily diffuse inside to the interface and feed the water splitting reaction so that a high current density can be applied to minimize the required membrane area. the Bipolar membranes have been successfully used in several commercial applications. They are currently developing other bipolar membranes for different applications.
There are used in conjunction with conventional monopolar membranes in several configuration: Three-Compartment configuration and Two-Compartment configuration.
The Three-Compartment configuration
A three-compartment cell is obtained by adding the bipolar membrane in a conventional electrodialysis cell. The bipolar membrane is flanked on either side by the anion- and cation-exchange membranes to form three compartments (see schematic below): acid between the bipolar and the anion-exchange membranes, base between the bipolar and the cation- exchange membranes, and salt between the cation- and anion-exchange membranes. As in ED stacks, many cells can be installed in one stack and a system of manifolds feeds all the corresponding compartments in parallel, creating three circuits across the stack: acid, base, and salt.

Schematic of Three-Compartment stack
It becomes easy to see how, by feeding the salt solution to the salt compartments, water to the acid and base compartments, and by supplying a DC current across the electrodes, it is possible to convert an aqueous salt solution such as NaCl into the base NaOH and the acid HCl. Similarly, other salts such KF, Na2SO4, NH4Cl, KCl, etc., as well as the salts of organic acids and bases can be converted.
The Two-Compartment configuration
There are two other main configurations that can be commonly considered: two-compartment cells with bipolar and cation-exchange membranes (only) or with bipolar and anion-exchange membranes. Using either two-compartment configuration might be only feasible in some cases and bring economic benefits such as lower investment costs and a lower operating cost.
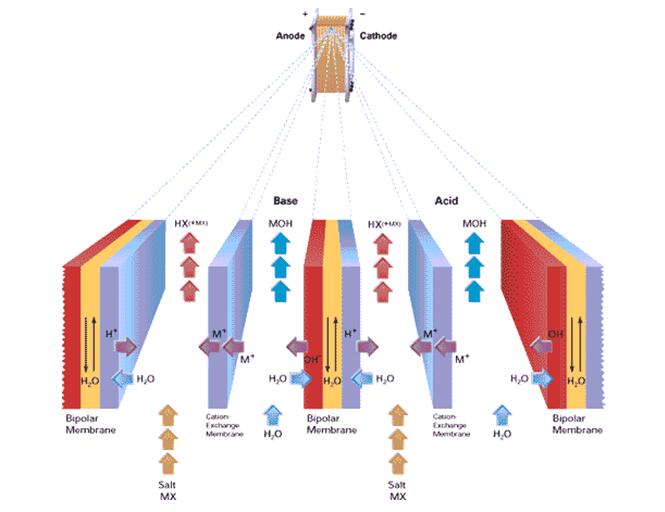
Schematic of Two-Compartment stack with cation-exchange membranes
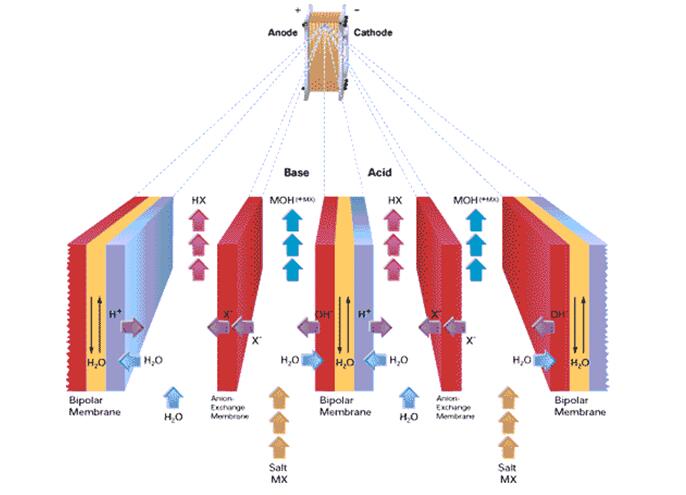
Schematic of Two-Compartment stack with anion-exchange membranes
The two-compartment cells with bipolar and cation-exchange membranes only are useful to convert the salts of weak acids and strong bases, such as sodium acetate, lactate, formate, glycinate, etc. and of other organic and amino acids. It also can be used in pH adjustment of Wine and Grape Juice
Similarly, the two-compartment cells with bipolar and anion-exchange membranes only are useful to convert the salts of weak bases (ammonia) and strong acids, such as ammonium chloride, ammonium sulfate, and ammonium lactate.
Copyright @ 2020-2024 Xiamen eSep Membrane Technology Co., Ltd. +86-592-5131 569 info@esep.co 闽ICP备16003752号
